OncoDNA
About OncoDNA
OncoDNA is a company specializing in precision medicine for the treatment of cancer and other genetic diseases. They are a group of passionate people who have developed a unique portfiolio of smart and personalized solutions to help users see clear through molecular complexity. Their customers can rely on the extensive expertise of the OncoDNA team in genomics and theranostics to collect, explore and interpret the tons of complex data that they need for their research project, clinical trial or clinical decision-making. OncoDNA aims to bring therapeutic innovation to the clinics and aid in delivering the best possible care to patients.
OncoDEEP: The best in class biomarker testing solution for tumor tissues
What is the Oncodeep kit?
The OncoDEEP Kit is a next-generation sequencing (NGS) assay that enables the in-house comprehensive genomic profiling of tumor samples. It was designed by oncology experts to contain the most relevant and complete cancer gene panel, and it has been extensively used and trusted by oncologists for 15 years. The OncoDEEP Kit is now available as an end-to-end solution for use in your lab. With it, you get rapid and reliable insights on a large number of complex cancer biomarkers in a single test.
Combined with fully automated data analysis and clinical interpretation software tools, the OncoDEEP Kit enables you to identify a broader set of treatment options for patients, while reducing on hands-on time and cost of outsourcing. The OncoDEEP Kit leverages the power of OncoDNA’s proprietary knowledgebase OncoKDO which counts no less than 4,500,000+ genetic variants, 1,400 drugs, 22,000+ documented genes, and data from more than 9,000 recruiting clinical trials. The kit is available in both RUO and CE-IVD versions.
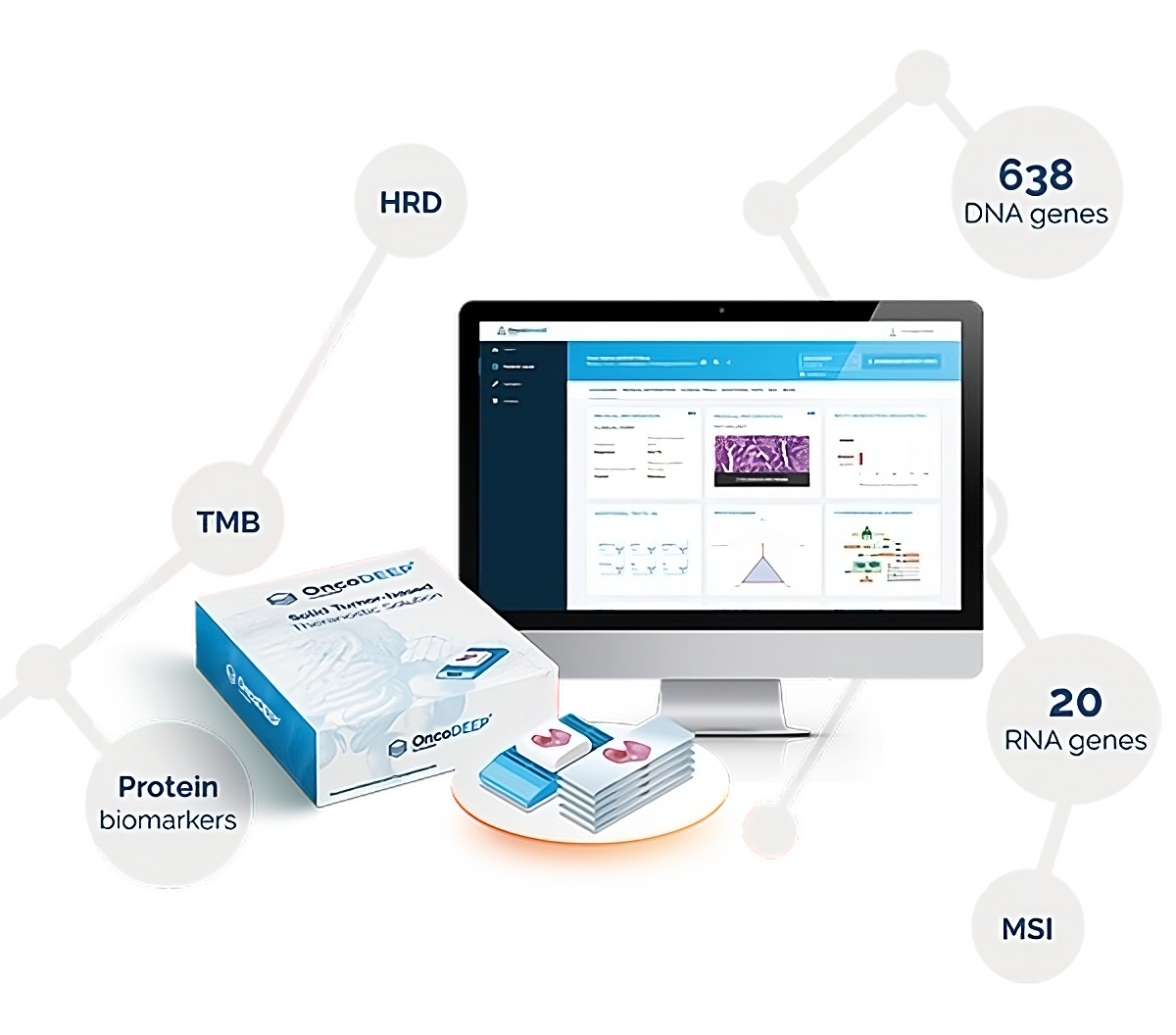
The technology behind the OncoDEEP kit
The OncoDEEP Kit supports the identification of all relevant DNA variants implicated in various solid tumor types with genes carefully selected based on their biological and therapeutical relevance. OncoDEEP DNA panel is composed of probes targeting 638 DNA genes and 20 RNA genes. In addition, specific sequences have been added to cover key regions associated with phenotypes of interest like tumor mutational burden (TMB), microsatellite instability (MSI), loss of heterozygosity (LOH) in tumor suppressor genes (TSGs), introns tilling for genes ALK/ROS1/RET and MET-ex14, sub-telomeric single-nucleotide polymorphisms (SNPs) for homologous recombination deficiency (HRD) calculation and the promotor of TERT.
The OncoDEEP Kit has been validated with formalin-fixed, paraffin embedded (FFPE) samples and has shown to achieve highly confident results with high sensitivity and specificity, thanks to Twist hybrid-capture chemistry using enzymatic fragmentation and unique dual indexes (UDIs), Illumina sequencing by synthesis (SBS) sequencing technology, and OncoDNA's sophisticated bioinformatics. The complete NGS workflow takes users from sample preparation to NGS sequencing and data analysis in just 4-5 days.

The OncoDEEP kit for HRD asessment
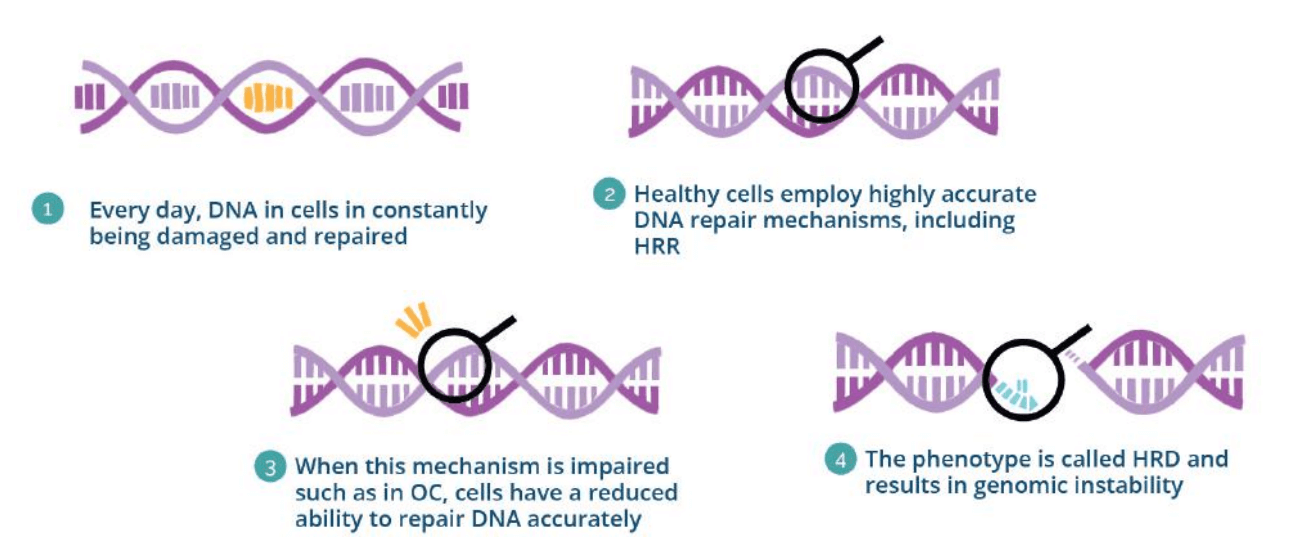
The Homologous Recombination Repair (HRR) pathway repairs DNA double-strand breaks (DSB). HRR deficiency leads to the accumulation of genomic aberrations and genomic instability which can promote malignant transformation. This phenotype of loss of the HRR capability and the associated genomic instability is called Homologous Recombination Deficiency (HRD). This phenotype has been associated with several cancer types, HRD status can thus provide crucial information about numerous cancer types, including ovarian, breast, pancreatic and prostate cancer. The utility of HRD in cancer treatment lies in its ability to predict response to specific types of therapy, as tumors with HRD are often more sensitive to DNA-damaging agents, such as platinum-based chemotherapy and PARP inhibitors. The OncoDEEP Kit is a ready-made solution can that can be used to report on HRD signature and get the most accurate results.
Our Knowledge in Your Lab
All relevant insights for in-house use
638 genes
4 classes of genomic alterations Complex genomic signatures (incl. HRD, MSI & TMB)
Control
Complete control over your data through software for detailed data exploration and clinical interpretation.
Superior efficiency
Thanks to the combination of OncoDNA’s proven expertise in oncology and Twist Bioscience’s library preparation and capture methods
Fast
From extracted DNA to clinical report in just 5 days Possibility to reduce hands on time through automation.
Trustworthy
(now version 7)
Used in clinical practice since 2014
For more details,
feel free to download
our following materials
Get a quote
Fill out the form and our colleague will contact you!
